  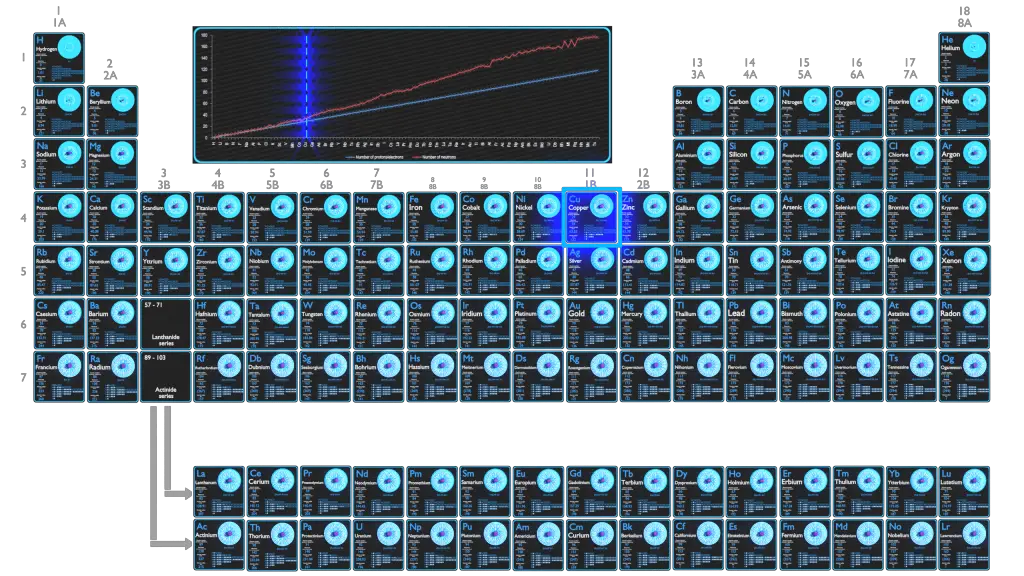
How many electrons and protons are in cu-63?Ģ9 protons, 29 electrons The number of neutrons depends on the isotope of copper. The number of electrons in an electrically-neutral atom is the same as the number of protons in the nucleus. Copper-65 is composed of 29 protons, 36 neutrons, and 29 electrons. How many protons, neutrons and electrons does copper have?Ĭopper-63 is composed of 29 protons, 34 neutrons, and 29 electrons. Both isotopes have 29 protons, so copper-63 has 34 neutrons and copper-65 has 36 neutrons. The two isotopes have different atomic masses. A heavy metal trace element with the atomic symbol Cu, atomic number 29, and atomic weight 63.55. What is copper 63 mass number?Ĭopper-63 is the stable isotope of copper with relative atomic mass 62.929601, 69.2 atom percent natural abundance and nuclear spin 3/2. The mass number of the atom (M) is equal to the sum of the number of protons and neutrons in the nucleus. The number of electrons in a neutral atom is equal to the number of protons. The number of protons in the nucleus of the atom is equal to the atomic number (Z). 
What’s the number of protons in copper?Ĭopper/Atomic number How do you find the number of protons electrons and neutrons in an element? Therefore, an atom of 63Cu contains 29 protons, 29 electrons and 34 neutrons. How many protons neutrons and electrons are in the following isotope of copper 63cu? The 65Cu isotope has 65 – 29 = 36 neutrons. How many neutrons does the isotope copper 65 have copper has an atomic number of 29? This means that the copper atom has 29 protons and 34 neutrons in its nucleus (29 + 34 = 63)…. How many protons neutrons and electrons does copper 63 have? In an unbalanced atom, the number of electrons equals the number of protons plus the opposite of the ion charge. In a balanced atom, the number of electrons equals the number of protons. The atomic number equals the number of protons. Using the Periodic Table, find the atomic number of the element. How do you find the protons neutrons and electrons of an isotope? The neutron has a mass very similar to that of the proton, so the two isotopes differ in mass. The two different kinds of atom are called isotopes of copper. One has 29 protons and 34 neutrons in the nucleus the other has 29 protons and 36 neutrons (Figure 4). In natural copper, the atoms are of two kinds. How many neutrons are in the isotope of copper? 9 How many electrons and neutrons does chromium have?.8 How many isotopes of copper have the same atomic number?.7 How many electrons and protons are in cu-63?.5 How do you find the number of protons electrons and neutrons in an element?.4 What’s the number of protons in copper?.3 How many protons neutrons and electrons does copper 63 have?.2 How do you find the protons neutrons and electrons of an isotope?.1 How many neutrons are in the isotope of copper?. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
